Camille’s medical records filled three drawers full of documentation. Source: www.afj.org
Like many pre-teens, 12-year-old Camille Baruch of Maryland had just one major thing to worry about — acne. When her doctor prescribed a pharmaceutical called Isotretinoin to treat it, Baruch and her parents and could not have imagined that the drug would cause her to develop ulcerative colitis, a severe chronic inflammatory bowel disease, which led to multiple surgeries — and eventually death by “unknown cause” at the age of 19.
What outraged many is that because Isotretinoin is a generic drug and not a brand name drug, its manufacturer cannot be sued by federal law, and is not liable for the injury.
It’s a very serious problem that Camille and her family learned firsthand, and one that is still not being given a fraction of the attention that it deserves in today’s day and age where most people don’t understand the risks of generic drugs.
Generic Drugs’ Side Effects Swept Under the Rug
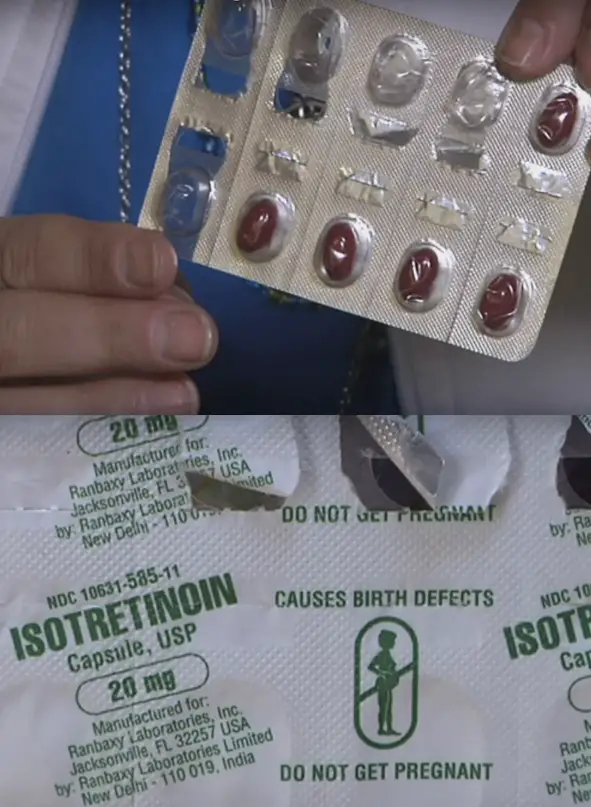
Isotretinoin is a generic form of Accutane, a drug that was commonly used to treat severe acne between 1982 and 2009. Accutane has been linked to another severe bowel disorder — Crohn’s disease (as well as birth defects and suicide), and it was removed from the market after thousands of lawsuits emerged (although the manufacturer Hoffmann-LaRoche, a part of The Roche Group, claims it was due to falling sales).
The company has paid over $53 million to Accutane patients for causing their Crohn’s disease, miscarriages, birth defects of their children, depression, and suicide.
But because Isotretinoin is a generic “no brand,” it is freely available on the market without proper warnings about all of its side effects. The only side effect that it openly discusses is birth defects. When Camille’s doctor and his medical staff discussed the drug the following is the only warning she received:
“The biggest thing they stressed was to not get pregnant. They didn’t mention anything else about any other side effects,” Camille said.
Camille’s Story: From Acne to Deliberating Pain and Eventually Early Death

Because of the bowel disorder caused by the acne medication, Camille had to take a dozen different medications. Source: www.afj.org
After Camille developed severe gastrointestinal problems, her life became a endless quest of searching for a way to stop the horrible pain. The symptoms just kept getting worse until the doctors decided to do surgery, the first one out of nine she would go through before her passing. The first took out her whole large intestine. But the surgeries did not solve the problem.
“Camille had every complication in the book,” her mom said.
Unfortunately for Camille, the only solution offered was to take more and more drugs — which led to still more painful side effects. Near the end of her life, Camille was taking a dozen different medications such as drugs for spasms, as well as antibiotics. Her condition would lead to a lifetime of “debilitating health problems” before Camille sadly passed away unexpectedly.
“She came home Friday night, took the medicine prescribed to her, went to bed Friday night and never woke up. It was an unbelievable shock,” her mom said.
Both during her life and now after, her parents, her doctors, and her lawyers have all been powerless to sue the maker of the drug that destroyed her health. And to blame is a 2011 Supreme Court decision that provided legal protection for generic drugs in cases like Camille’s.
PLIVA v. Mensing: The Court Case that Removed Liability from Generic Drug Makers
Lately, more and more Supreme Court Cases end up protecting the interests of the corporations instead of consumers.
“In case after case, a solid 5-vote conservative majority on the Supreme Court has begun to tip the balance towards corporate interest,” Editor and Publisher of The Nation Katrina vanden Heuvel said.
Such is the case of PLIVA v. Mensing.
The case began after Gladys Mensing sued PLIVA, a pharmaceutical company, after a generic anti-nausea drug Reglan led her to develop a severe neurological movement disorder. Gladys claimed that PLIVA did not properly warn consumers of this health risk on its labels.
PLIVA claimed that they were following the federal law that requires generic drug labels to be identical to brand name labels.
The Supreme Court decided that generic drugs are not liable even when refusing to tell the FDA about new health risks that were discovered. They only need to copy the warning signs that the brand name drugs have. Thus these drugmakers have “no control over what their labels say” and cannot be sued when their products cause serious illness or even death.
According to this article from The New York Times, stacks of lawsuits are being dismissed in federal court because of this little known detail.
“Your pharmacists aren’t telling you, hey, when we fill this with your generic, you are giving up all of your legal remedies,” said Michael Johnson, Mensing’s lawyer, in the Times. “You have a disparate impact between one class of people and another.”
Since generic drugs make 75% of the drug market, this becomes a serious issue, especially in regards to consumers like Camille whose lives were already destroyed by these drugs.
“In the PLIVA case the Supreme Court ruled that federal law overrides the state laws. Federal law announces — no, the generic company has no responsibility if their drug has injured a consumer, and it’s a very serious problem.”
Although the court case was about Regla, this decision affects every generic drug.
Before her passing an ailing Camille was interviewed by Alliance for Justice to spread awareness of what happened to her.
“It hurts me…knowing that no one is going to be held responsible for this… My life has been shattered… But knowing that other people are getting sick from it is just as bad,” she said.
You can see the full video below (and feel free to share this article with someone who doesn’t know the risks associated with generic drugs):
Thanks for installing the Bottom of every post plugin by Corey Salzano. Contact me if you need custom WordPress plugins or website design.